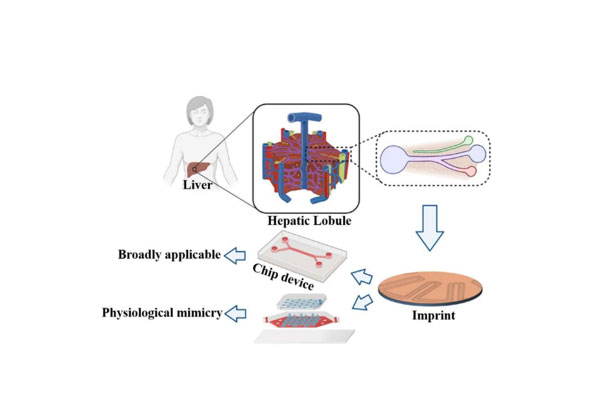
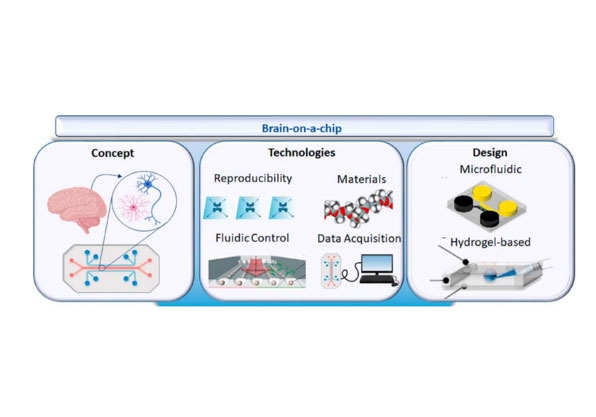
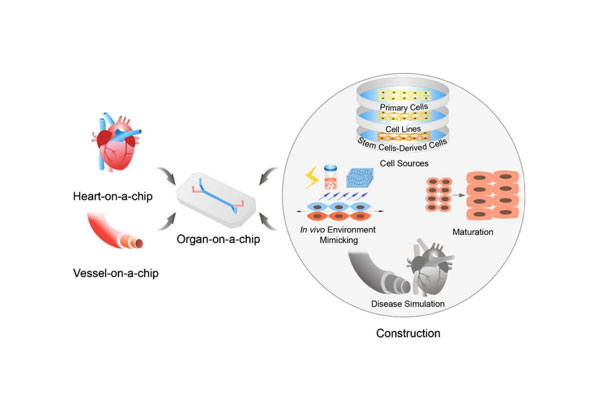
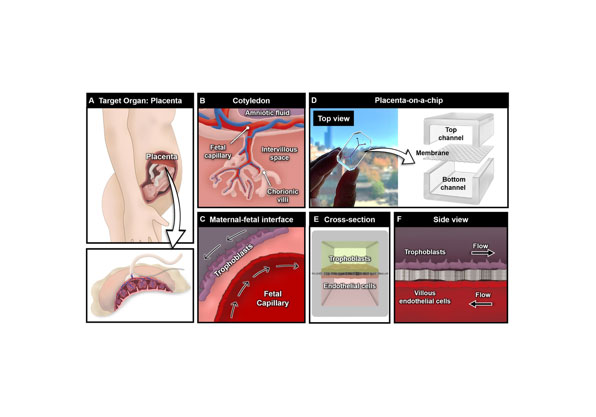
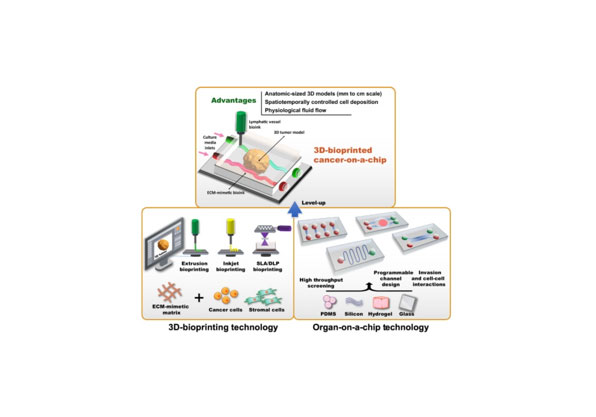
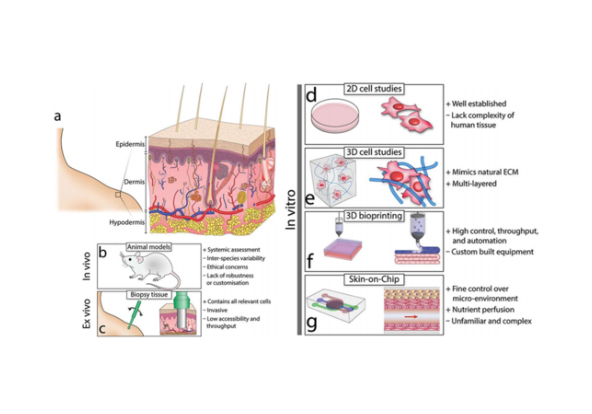
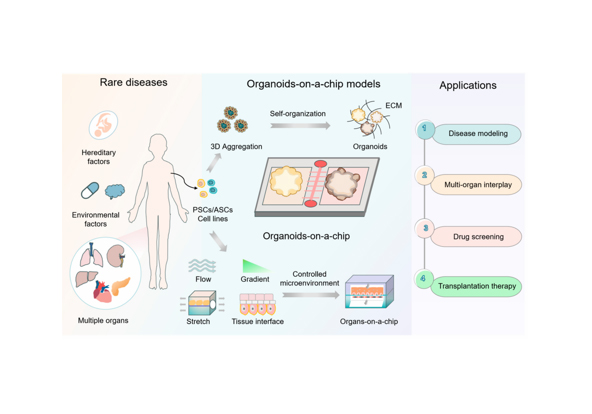
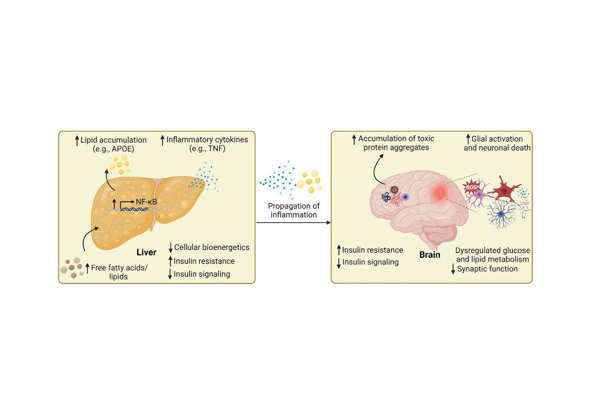
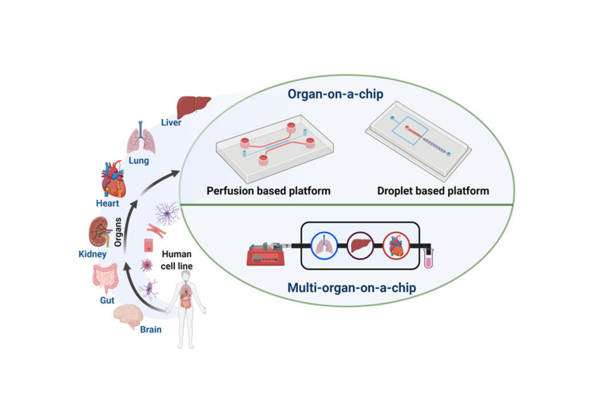
Liver
Liver-on-a-Chip constructs a three-dimensional microenvironment encapsulating hepatocytes and non-parenchymal cells (e.g., Kupffer cells, hepatic stellate cells) on a microchip platform, thereby recapitulating the physiological structure and functional characteristics of the human liver. This in vitro model provides a robust experimental tool for systematic investigations into liver function regulation, drug-induced toxicity evaluation, drug metabolic profiling, and the pathogenic mechanisms underlying liver diseases.